A 78-year old patient presented with worsening exertional dyspnea and pedal edema.
His history included arterial hypertension, hyperlipidemia, atrial fibrillation and atrial flutter (treated with catheter ablation) and 2nd degree AV block Wenckebach type documented on Holter monitoring at night.
Figure 1 ECG (on admission) Atrial fibrillation with slow ventricular response (HR 65/min), diffuse low voltage QRS complexes, “pseudo-infarction” Q waves (III, aVF, V1-V3)

Echocardiography examination showed non-dilated left ventricle with moderate systolic dysfunction (EF=35-40%). Diffuse hypokinesis with maximum wall motion abnormalities in the interventricular septum and the inferior wall of the left ventricle. Concentric hypertrophy of the LV including the inter ventricular septum (13-14mm) was observed. Significant diastolic dysfunction and bilateral atrial dilation was reported. Mild tricuspid regurgitation and small pulmonary regurgitation.
Video 1 Echocardiography examination
.gif)
Patient was started on anticoagulation therapy and diuretics. Cardiac magnetic resonance (CMR) and cardiac scintigraphy were scheduled to rule out suspected cardiac amyloid disease.
Results of CMR were as follows:
Left ventricle: EF 53 %, EDVi 75 ml/m2, ESVi 35 ml/m2, LV mass 113 g/m2 (normal size).
Right ventricle: EF 43 %, EDVi 95 ml/m2, ESVi 54 ml/m2 (normal size).
Late gadolinium enhancement of both ventricles with maximum changes in the interventricular septum = CMR pattern typical for cardiac amyloidosis.
Figure 2 Late gadolinium enhancement of both ventricles with maximum changes in the interventricular septum

Technetium-labeled cardiac scintigraphy, 99mTcDPD SPECT/CT and gated SPECT were performed: Technetium accumulation in both left and right ventricles with higher accumulation in the left ventricle. Highest activity noted in the interventricular septum and apex.
Figure 3 99mTcDPD SPECT/CT

DC cardioversion was successfully performed, however long term sinus rhythm was not achieved. Pedal edema subsided after diuretic therapy. Patient is currently classified as heart failure NYHA II class. The diagnosis of TTR amyloidosis was confirmed and the patient is currently a participant of trial Apollo-B (Patisiran vs. placebo).
Clinical context
Transthyretin amyloidosis is emerging as an underdiagnosed cause of heart failure especially in the older population. Age at onset is 50–60 years with average life-expectancy of 7–14 years. Recent studies suggest that up to 10-15% of older patients with heart failure may have unrecognized wtATTR. Another recent study has shown that 8% of patients undergoing TAVR had TTR amyloidosis.
There exist two types of TTR amyloidosis:
a) a hereditary form caused by gene mutation
b) a wild type TTR amyloidosis that is much more common and in which the transthyretin becomes. unstable during ageing.
TTR amyloidosis affects mainly the heart where it accumulates, but it can also cause neuropathy (peripheral and/or autonomic).
TTR amyloidosis manifests as an infiltrative cardiomyopathy and heart failure with its signs and symptoms. Other possible symptoms include arrhythmias and conduction system disease.
Learning points
1) In older patients, heart failure may be caused by TTR amyloidosis (usually wild type). Up to 15% of older patients with HF may have TTR amyloidosis. Populations of patients with aortic stenosis referred for TAVR are demographically similar to patients with TTR amyloidosis.
2) A history of HF with carpal tunnel syndrome, lumbar spinal stenosis, biceps tendon rupture or polyneuropathy should raise the suspicion for cardiac amyloidosis. In our case, the patient had a history of lumbar spinal stenosis. Lumbar spinal stenosis also presents in late middle-age; related to disc degeneration, spondylolisthesis and age-related degenerative processes, such as WT-TTR amyloid deposition.
3) Diagnosis of TTR amyloidosis may begin with echo and/or CMR, however nuclear imaging and ruling out the possibility of AL amyloidosis is needed to confirm the diagnosis, while endomyocardial biopsy is no longer required.
Echo signs suggestive of amyloidosis are:
- Increased LV wall thickness with/without right ventricular wall thickness
- Decreased longitudinal strain with apical sparing
- Other classic echocardiographic signs of cardiac amyloidosis are biatrial dilatation, mild pericardial. effusion, atrioventricular valve thickening, atrial septal wall thickening, and granular sparkling. appearance of the myocardium.
4) Cardiac imaging can replace endomyocardial biopsy if it is performed using Tc-99m- PYP, Tc-99m−3,3-diphosphono-1,2-propanodicarboxylic acid (DPD) or Tc-99m−hydroxymethylene diphosphonate (HMDP) - their common feature is bone avidity. At the same time, testing for the absence of a monoclonal protein by serum and urine testing and features suggesting TTR amyloidosis on echo or CMR are required.
5) There are typical ECG features related to restrictive cardiomyopathy associated with amyloidosis:
- Diffuse myocardial infiltration leads to low voltage QRS complexes.
- Atrial fibrillation may occur due to atrial enlargement; ventricular arrhythmias are also common.
- Infiltration of the cardiac conduction system (e.g. due to septal granuloma formation in sarcoidosis) may lead to conduction disturbance — e.g. bundle branch blocks and AV block.
- Healing granulomas in sarcoidosis may produce “pseudo-infarction” Q waves.
- Novel causal therapy for cardiac TTR amyloidosis is currently being evaluated - tafamidis. Treatment of HF should be initiated if it is present as is the case if arrhythmias are present.
References
- Braunwald's Heart Disease: A Textbook of Cardiovascular Medicine, 11th ed, Zipes DP, Libby P, Bonow RO, et al, W.B. Saunders Company, Philadelphia 2018.
- Hanna, Mazen, Frederick L. Ruberg, Mathew S. Maurer, Angela Dispenzieri, Sharmila Dorbala, Rodney H. Falk, James Hoffman, et al. 2020. “Cardiac Scintigraphy With Technetium-99M-Labeled Bone-Seeking Tracers For Suspected Amyloidosis”. Journal Of The American College Of Cardiology 75 (22): 2851-2862. https://doi.org/10.1016/j.jacc.2020.04.022.
- Ruberg, Frederick L., Martha Grogan, Mazen Hanna, Jeffery W. Kelly, and Mathew S. Maurer. 2019. “Transthyretin Amyloid Cardiomyopathy”. Journal Of The American College Of Cardiology 73 (22): 2872-2891. https://doi.org/10.1016/j.jacc.2019.04.003.
- https://litfl.com/restrictive-cardiomyopathy-ecg-library/
Authors: Michal Pazderník, Michael Jenšovský
You Might Also Like
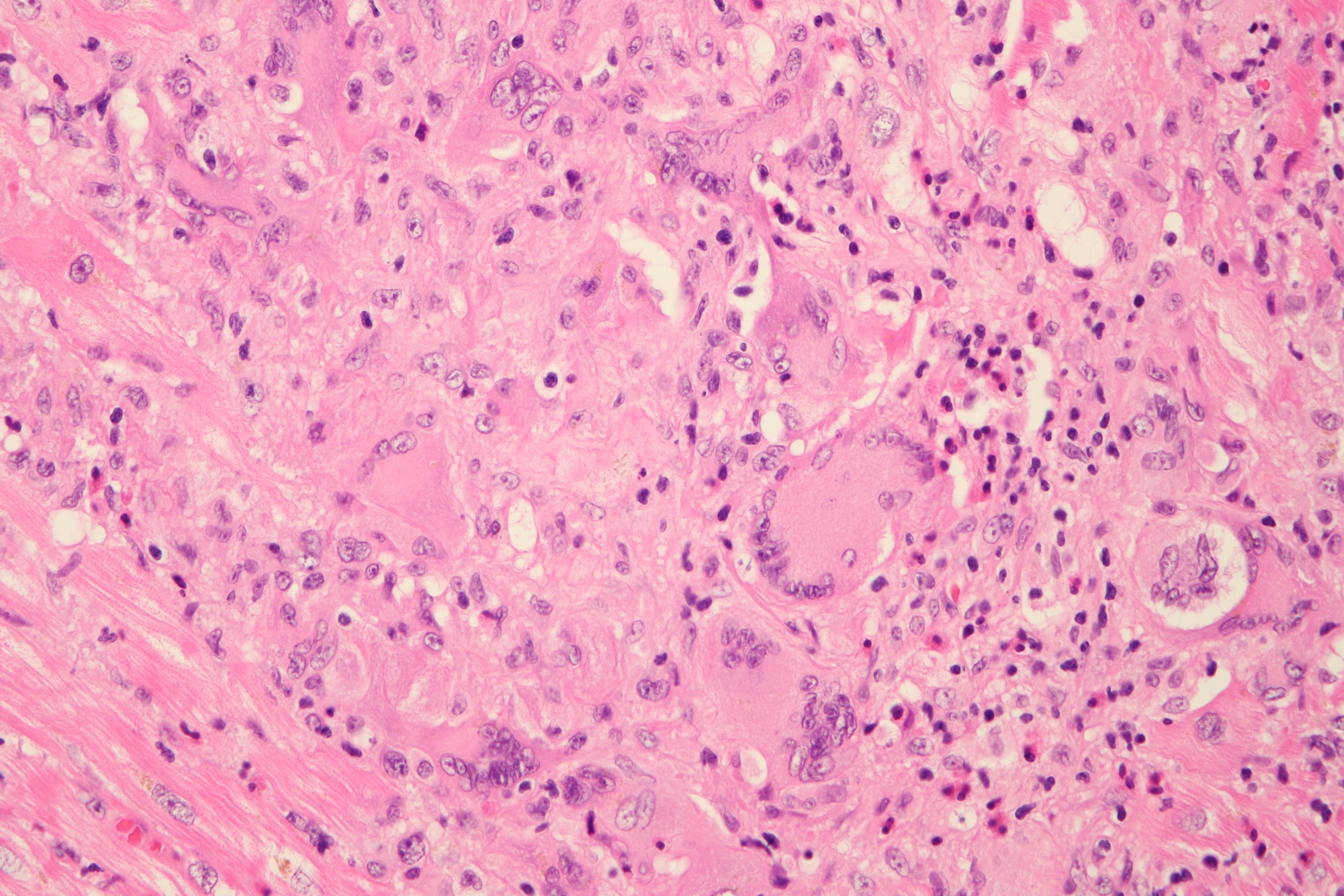
Cardiogenic shock due to Giant Cell Myocarditis
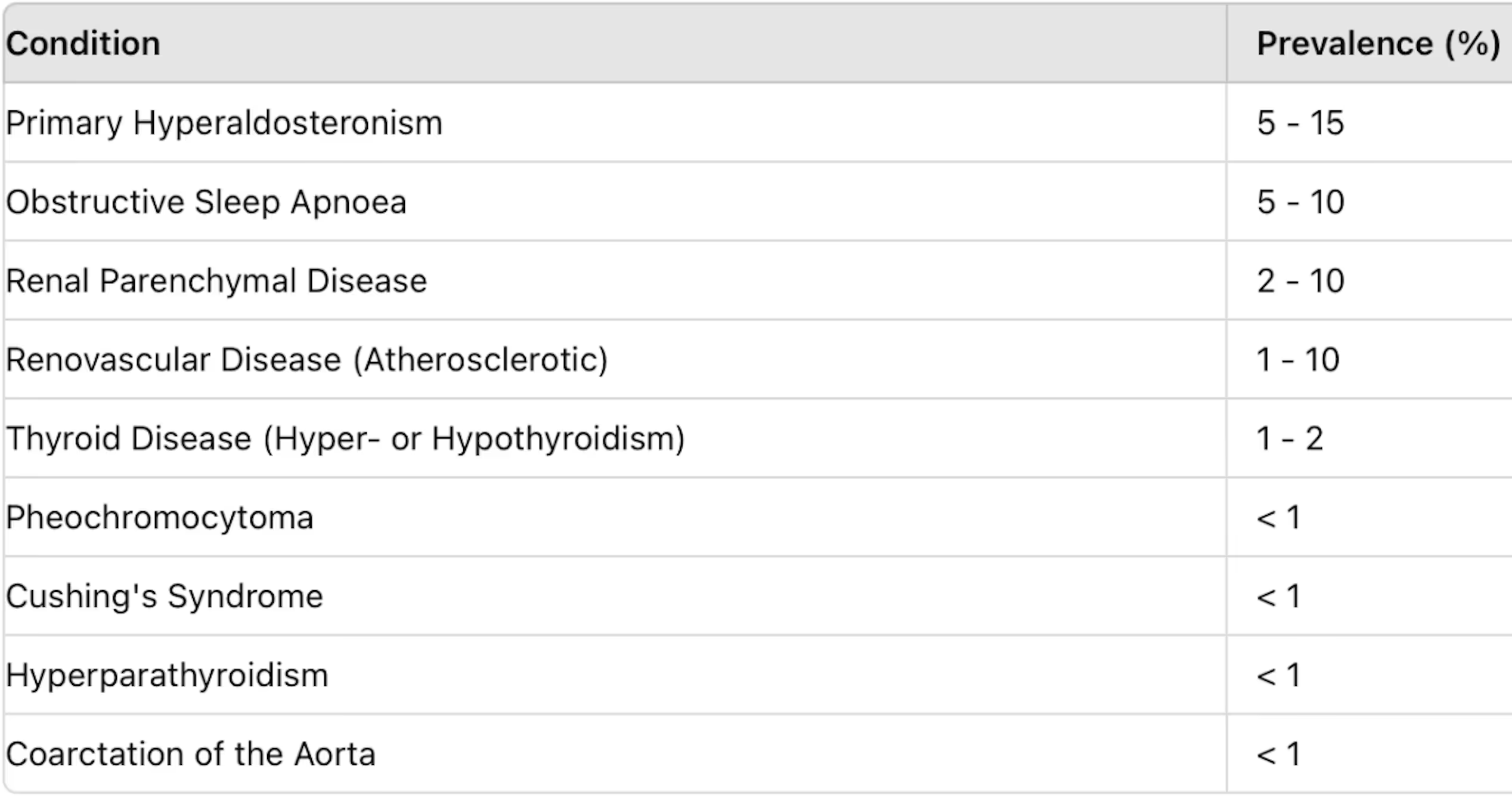
Secondary hypertension


.jpg)


