
Thrombotic complications in a patient with COVID-19 pneumonia
A 55-year-old patient with a history of systemic hypertension, dyslipidemia, COPD and emphysema was admitted to a hospital for three days persiting fever and chest pain. The patient had known contact with COVID-19 positive person one week ago. Laboratory findings showed peak of hsTnT (440 ng/l) and elevated levels of CRP (171 mg/l). ECG revealed signs of STEMI inferior accompanied with AV block III.type.
Figure 1 – ECG on admission, ST elevations in II, III, aVF, ST depressions I,a VL, 3rd degree AV block.

Urgent selective coronarography was performed and thrombotic obliteration of the middle part of the right coronary artery (RCA) was found. Following thrombus extraction and eptifibatide administration, PCI with DES implantation was performed.
Video 1 Thrombotic occlusion of right coronary artery

After PCI, sinus rhythm was restored and all remaining clinical symptoms were associated with COVID-19 bilateral pneumonia (confirmed via a PCR test) with suspected bacterial superinfection (CRP 268 mg/l, leukocytosis 22.8 10^9/l, ). Fibrinogen levels were 7.05 g/l.
CT of the chest showed bilateral pneumonia with possible post-pneumonia changes in chronic emphysema terrain.
Figure 2 CT chest with signs of COVID pneumonia

CT angiography excluded pulmonary embolism (PE), but thrombi in the descending aorta and aortic arch were found. This thrombotic complication was most probably associated with hypercoagulative state caused by SARS-CoV-2 infection.
Figure 3 Thrombi present in aortic arch and descending aorta

Empiric ATB therapy with levofloxacin + cefotaxime was commenced, and anticoagulation therapy was started. Patient rapidly improved within the next few days, and was stable on a non-invasive oxygen therapy.
Clinical context
There is undeniable evidence that there is a significant risk of thrombosis in patients with COVID-19. A coagulopathy has been reported in up to 50% of patients with severe COVID-19 manifestations. The most significant change in coagulation parameters is an increase in D-dimers and progressively increasing values can be used as a prognostic parameter.
The most frequent thrombotic complications include pulmonary embolism (PE) or pulmonary thrombosis (PT), deep-vein thrombosis (DVT), ischaemic stroke, myocardial infarction and systemic arterial embolism.
Most studies are exploring this phenomenon in patients with severe course of the disease, who are admitted to hospital and often need intensive care.
The SARS-CoV-2 virus is connected with a hypercoagulative state that is characterised by derangements in clotting tests (PT and APTT), elevated D-dimers and an increased thrombotic status. Hypercoagulation is triggered by multiple factors. Recent studies showed a complicated process that involves direct interaction of the virus with particular tissues and platelets, inflammation, complement activation and ineffective thrombolysis. Nevertheless, it is yet unknown whether these hemostatic changes are a specific effect of SARS-CoV-2 or are a consequence of cytokine storm that precipitates the onset of SIRS, as observed in other viral diseases.
Figure 4 Mechanisms of coagulopathy and thrombosis in COVID-19

Eden M.PageRobert A.S.Ariëns, . Mechanisms of Thrombosis and Cardiovascular Complications in COVID-19, Elsevier, 2021, https://ars-els-cdn-com.ezproxy.is.cuni.cz/content/image/1-s2.0-S0049384821000141-gr1.jpg
Proposed mechanisms of immunothrombosis in COVID-19 and the interactions of the inflammation and coagulation systems. Direct SARS-CoV-2 – platelet interaction results in high levels of platelet activation, promoting a prothrombotic state.
The SARS-CoV-2 potentiates a hyper-coagulable state generally in 4 ways:
- Activation of the contact and tissue factor pathways of coagulation
- The virus causes direct myocardial and microvascular injury that results in subendothelium and collagen exposure.
- The endothelial injury is mediated by the interaction of the virus with angiotensin converting enzyme 2 (ACE2) receptor that enables the virus to enter the cells. ACE2 receptors are widely expressed in tissues such as lung epithelium, myocardium, kidneys and vascular endothelium.
- Tissue factor (TF) is exposed in the subendothelium and activates the tissue factor pathway by FVII cleavage.
- The trauma also contributes to platelet activation and subsequent intrinsic way activation.
- Interactions between SARS-CoV-2 and platelets
- SARS-CoV-2 induces functional changes in platelets by yet not fully understood mechanisms, but platelet uptake of the virus is suspected.
- Platelet hyperreactivity causes increased platelet-platelet and platelet-leukocyte interactions and potentiates degranulation with polyphosphate release from activated dense granules.
- This results in activation of the intrinsic pathway via the activation of FXII and in the end - fibrin polymerisation.
- Inflammatory mediators production
- As a response to the infection, inflammatory mediators such as C-Reactive protein (CRP), Interleukin 6 (IL-6), Interleukin 8 (IL-8) and tumour necrosis factor α (TNF-α) are produced.
- IL-6 is a key regulator of fibrinogen transcription. Drastic rise in plasma fibrinogen is observed in COVID-19 patients with increased IL-6 levels.
- The release of neutrophil extracellular traps (NETs) from neutrophils
- Evidence suggests increase of NETosis in patients with severe COVID-19. NETosis is an important part of immune response that traps viruses, fungi and bacteria, whilst concentrating anti-microbial factors.
- Increased NETosis interacts with inflammatory and coagulation cascades, which contributes to acute lung injury and endothelium, platelet and FXII activation.
- The presence of NETs also increases the resistance of clots to fibrinolysis. All this worsens the pro-coagulative state.
Figure 5 Mechanisms of coagulopathy and thrombosis in COVID-19

Bikdeli at al., B. “Postulated Mechanisms of Coagulopathy and Pathogenesis of Thrombosis in COVID-19.” PubMed Central, 2021, www.ncbi.nlm.nih.gov/pmc/articles/PMC7164881/figure/undfig2/.
The result of hypercoagulative state created by SARS-CoV-2 shows specific damage that is not typical in other similar viral infections.
Pulmonary micro-thrombosis – ultrasound-based autopsy techniques revealed fibrin thrombi in the pulmonary arterioles in 8 out of 10 cases. This indicates primary pulmonary thrombosis that is most probably causing the typical rapid development of hypoxemia in the early stages of ARDS in COVID-19 patients. The microthrombi are creating a ventilation/perfusion mismatch and subsequent dead space enlargement.
Pulmonary thrombosis rather than embolism – Diagnosis of pulmonary embolism is the most common outcome of hypercoagulative state in COVID-19 patients and it appears that in many cases PT is not always preceded by deep-vein thrombosis (DVT) and more likely originates primarily from the lungs. This is consistent with the pulmonary micro-thrombosis findings and has two practical consequences. 1) the commonly used Well’s criteria for PE that depends on presence of DVT symptoms is no longer relevant scoring system and 2) the PT has different pathogenic mechanisms than PE and may alter treatment recommendations. Standard prophylactic therapy with LMWH may not be sufficient to prevent pulmonary thrombotic complications.
Disseminated intravascular coagulation – DIC incidence has been reported in 2,2% in hospitalized patients, however, 71,4% of non-survivors meets International Society of Thrombosis and Haemostasis (ISTH) DIC criteria (which include platelet count, fibrinogen, D-Dimer and prothrombin time), compared with 0,6% of the survivors. The coagulopathy in COVID-19 appears to be distinct from DIC due to other causes. There is an apparent lack of bleeding in patients, only modest prolongation of the APTT, elevated fibrinogen and the absence of schistocytes on the blood film. There are several theories to explain this, from localized pulmonary intravascular coagulation stemming from endothelial cell ACE2 receptor interaction with SARS-CoV-2 to complement-mediated thrombotic microangiopathy with additional presence of anaemia, LDH elevation and renal dysfunction. Presence of DIC symptoms is therefore likely to be a useful prognostic tool.
Cardiovascular complications – acute cardiac injury has been observed in 7-28% hospitalized patients and is associated with an increased risk of severe disease and death. This may be due to virus-mediated lysis of cardiomyocytes (which is observed in other viral infections) or by binding to ACE2 receptors in the heart. When the virus binds to ACE2 receptors, the activity of ACE2 is reduced and may decrease cardioprotective effects of the enzyme. Myocarditis has been suspected in many patients, but the incidence is unknown due to incomplete histology tests.
Atherosclerotic plaque instability – inflammation and cytokine release increase inflammatory cell migration and plaque infiltration that can result in plaque instability with subsequent arterial thrombus formation. Atherosclerotic plaque rupture can lead to coronary thrombosis and acute myocardial infarction. Overall, patients with risk factors for atherosclerosis, such as hypertension and diabetes, are 2-3x times more likely to develop severe symptoms requiring ICU admission.
What markers to look for in COVID-19?
- D-Dimers - has been proposed as an important prognostic marker for mortality in COVID-19 disease. In a study of 150 patients with ARDS, >95% has elevated D-dimer levels. They could also be useful as a tool for anticoagulation monitoring, as D-dimer levels decrease after successful treatment. Reports suggest that 2 μg/ml (4x the reference range of 0,5μg/ml) could be used as a cut off level for pre-empting mortality. Therefore, serial monitoring of D-dimers can be a useful biomarker of clinical severity in patients with COVID-19. The more severe the clinical picture is, the higher values usually are.
- Platelet count – thrombocytopenia is a common finding in COVID-19 patients as a result of platelet consumption, destruction or decreased production. However, it rarely results in haemorrhagic complications (2,7%). Thrombocytopenia is associated with > 5 times increased risk of severe disease.
- Prothrombin time (PT) and Activated Partial Prothrombin Time (aPTT) – these are less specific prognostic markers and their change due to hypercoagulability is relatively small. Overall, PT tends to prolong (>3s) and aPTT is usually higher on admission and slowly decreases across the same period.
- Cardiac specific troponins T and I are consistently elevated in patients with COVID-19 and are also markers of poor prognosis as the elevation occurs frequently in ICU patients and non-survivors. It may be caused by myocardial injury, impaired renal function, myocarditis, PE or acute myocardial infarction.
- Interleukin 6 (IL-6) – a key regulator of fibrinogen transcription is responsible for drastic rise in plasma fibrinogen. IL-6 levels may correlate with disease severity and a procoagulant profile. There has also been considerable interest in the therapeutic potential of the IL-6 receptor antagonist, tocilizumab. Two separate case reports of the use of tocilizumab in acute chest syndrome of Sickle cell disease precipitated by COVID-19 reported rapid responses in an adult and a teenager.
- Fibrinogen, ferritin, CRP, LDH – all these inflammatory markers are also elevated and might take a role in creation of the hypercoagulative state.
Figure 6 Prophylaxis and therapy of COVID-19 associated coagulopathy

American College of Cardiology Foundation, . “Variability in Resources and Testing Strategies, and in Contracting COVID-19 After Exposure to SARS-CoV-2.” PubMed Central, 2021, www.ncbi.nlm.nih.gov/pmc/articles/PMC7164881/bin/gr1_lrg.jpg.
Management
- The backbone of the management of thrombotic complications is prevention as well as early recognition and treatment of thrombotic events.
- Studies suggest that prophylactic use of heparin reduces mortality in high-risk patients.
- Patients with an elevated d-dimer (arbitrarily defined as a 3- to 4-fold increase) should be admitted to hospital.
- Low molecular weight heparin (LMWH) at standard prophylactic doses should be considered in all patients who need to be hospitalized for COVID-19 infection unless contraindicated. It is the preferred anticoagulant agent as it does not interfere with PT or aPTT and has anti-inflammatory properties.
- Unfractionated heparin with antiXa monitoring may be a useful alternative as it is frequently used in intensive care. Look out for Heparin-Induced Thrombocytopenia (HIT).
- Microvascular thrombosis with widespread fibrin deposition reported in COVID-19 patients has led to clinical trials to evaluate the efficacy of experimental fibrinolytic therapies (tPA).
Interactions of anticoagulants with COVID-19 therapies
- No major interactions have been reported between investigational drugs for COVID-19 and heparin anticoagulation.
- Non-vitamin K antagonist oral anticoagulants (NOACs) may have interactions with some of the investigational drugs for COVID-19, notably lopinavir/ritonavir. In such cases, NOACs should be avoided.
- COVID-19 patients on known oral anticoagulation may be switched over to parenteral anticoagulation with LMWH and UFH when admitted to an ICU with a severe clinical presentation. This prevents these drug-drug interactions, except for azithromycin, which should not be co-administered with UFH.
Figure 7 Interactions of anticoagulant drugs with COVID-19 therapies

“Interactions of Anticoagulant Drugs with COVID-19 Therapies.” ESC Guidance for the Diagnosis and Management of CV Disease during the COVID-19 Pandemic, 2020, www.escardio.org/Education/COVID-19-and-Cardiology/ESC-COVID-19-Guidance
Figure 8 Drugs used for COVID-19 treatment and their interactions with commonly used cardiology drugs

„Potential interactions of drugs used to cure COVID-19“ ESC Guidance for the Diagnosis and Management of CV Disease during the COVID-19 Pandemic, 2020, www.escardio.org/Education/COVID-19-and-Cardiology/ESC-COVID-19-Guidance
References
- Page EM, Ariëns RAS. Mechanisms of thrombosis and cardiovascular complications in COVID-19. Thromb Res. 2021 Jan 18;200:1-8. doi: 10.1016/j.thromres.2021.01.005. Epub ahead of print. PMID: 33493983; PMCID: PMC7813504.
- Gabrielli M, Lamendola P, Esperide A, Valletta F, Franceschi F. COVID-19 and thrombotic complications: Pulmonary thrombosis rather than embolism? Thromb Res. 2020 Sep;193:98. doi: 10.1016/j.thromres.2020.06.014. Epub 2020 Jun 8. PMID: 32534328; PMCID: PMC7278638.
- Bikdeli B, Madhavan MV, Jimenez D, Chuich T, Dreyfus I, Driggin E, Nigoghossian C, Ageno W, Madjid M, Guo Y, Tang LV, Hu Y, Giri J, Cushman M, Quéré I, Dimakakos EP, Gibson CM, Lippi G, Favaloro EJ, Fareed J, Caprini JA, Tafur AJ, Burton JR, Francese DP, Wang EY, Falanga A, McLintock C, Hunt BJ, Spyropoulos AC, Barnes GD, Eikelboom JW, Weinberg I, Schulman S, Carrier M, Piazza G, Beckman JA, Steg PG, Stone GW, Rosenkranz S, Goldhaber SZ, Parikh SA, Monreal M, Krumholz HM, Konstantinides SV, Weitz JI, Lip GYH; Global COVID-19 Thrombosis Collaborative Group, Endorsed by the ISTH, NATF, ESVM, and the IUA, Supported by the ESC Working Group on Pulmonary Circulation and Right Ventricular Function. COVID-19 and Thrombotic or Thromboembolic Disease: Implications for Prevention, Antithrombotic Therapy, and Follow-Up: JACC State-of-the-Art Review. J Am Coll Cardiol. 2020 Jun 16;75(23):2950-2973. doi: 10.1016/j.jacc.2020.04.031. Epub 2020 Apr 17. PMID: 32311448; PMCID: PMC7164881.
- Bhanu Kanth Manne, Frederik Denorme, Elizabeth A. Middleton, Irina Portier, Jesse W. Rowley, Chris Stubben, Aaron C. Petrey, Neal D. Tolley, Li Guo, Mark Cody, Andrew S. Weyrich, Christian C. Yost, Matthew T. Rondina, Robert A. Campbell; Platelet gene expression and function in patients with COVID-19. Blood 2020; 136 (11): 1317–1329. doi: https://doi.org/10.1182/blood.2020007214
- Agbuduwe C, Basu S. Haematological manifestations of COVID-19: From cytopenia to coagulopathy. Eur J Haematol. 2020 Nov;105(5):540-546. doi: 10.1111/ejh.13491. Epub 2020 Aug 31. PMID: 32663356; PMCID: PMC7404736.
- Lippi G, Favaloro EJ. D-dimer is Associated with Severity of Coronavirus Disease 2019: A Pooled Analysis. Thromb Haemost. 2020;120(5):876-878. doi:10.1055/s-0040-1709650
- Lippi G, Plebani M. Laboratory abnormalities in patients with COVID-2019 infection. Clin Chem Lab Med. 2020 Jun 25;58(7):1131-1134. doi: 10.1515/cclm-2020-0198. PMID: 32119647.
- von Meijenfeldt FA, Havervall S, Adelmeijer J, Lundström A, Rudberg AS, Magnusson M, Mackman N, Thalin C, Lisman T. Prothrombotic changes in patients with COVID-19 are associated with disease severity and mortality. Res Pract Thromb Haemost. 2020 Dec 6;5(1):132-141. doi: 10.1002/rth2.12462. PMID: 33537537; PMCID: PMC7845083.
- “ESC Guidance for the Diagnosis and Management of CV Disease during the COVID-19 Pandemic.” European Society of Cardiology, 2021, www.escardio.org/Education/COVID-19-and-Cardiology/ESC-COVID-19-Guidance.
Authors: Michal Pazdernik, Lucie Mayerová
You Might Also Like
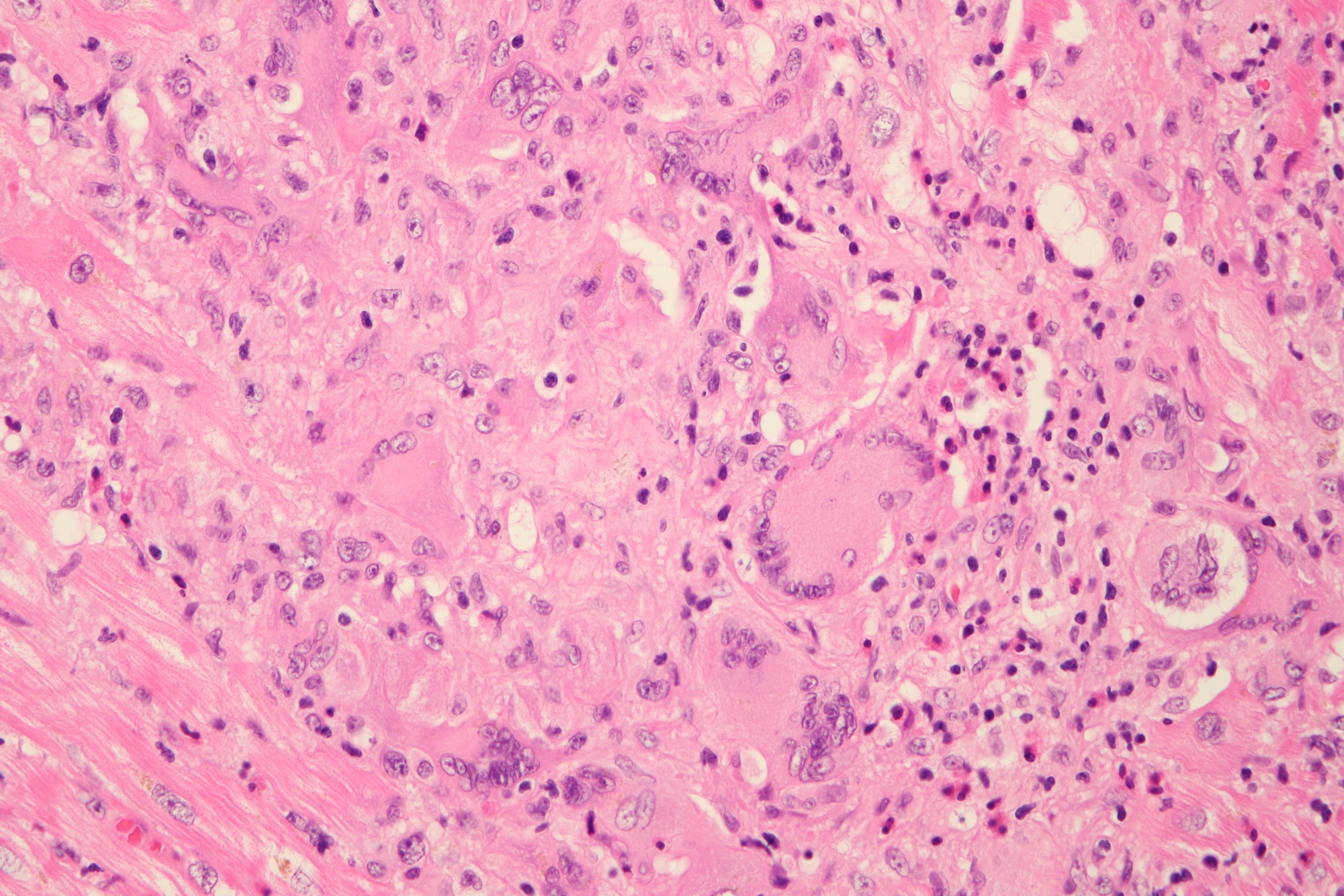
Cardiogenic shock due to Giant Cell Myocarditis
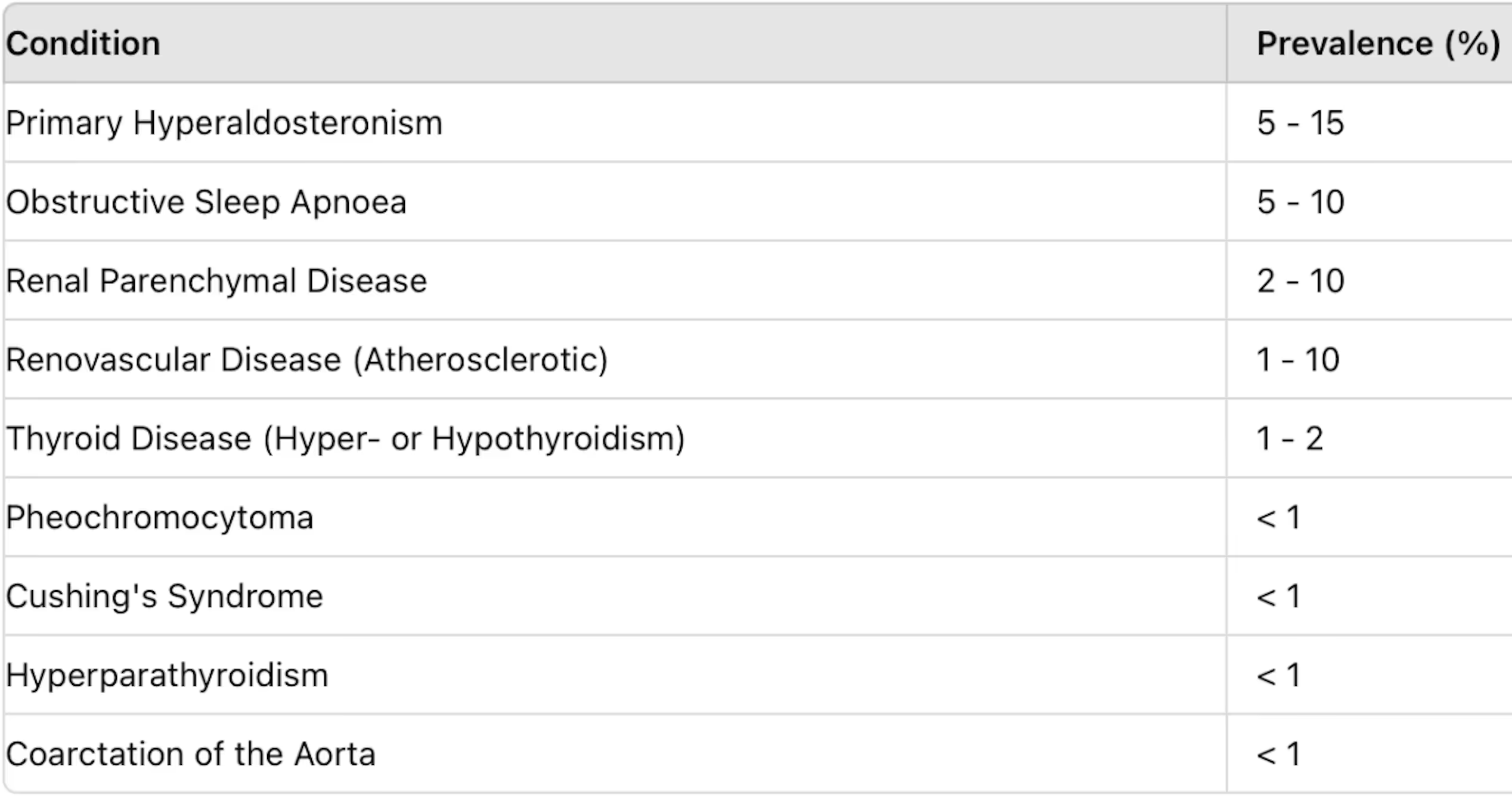
Secondary hypertension

.jpg)


