A 46-years old patient with a history of deep vein thrombosis (heterozygous for Leiden mutation), was admitted for newly documented chest pain with irradiation to neck and under left scapula. Laboratory findings shower mild elevation of cardiac-specific enzymes (hsTnT 19 ng/l, normal range < 14) and D-dimers (2 mg/L, normal range <0,5).
ECG on admission showed no signs of acute coronary syndrome.
Figure 1 ECG on admission

Transthoracic echocardiography showed normal function of both ventricles, presence of circumferential pericardial effusion (maximum approx. 8 mm around RV), ascending aorta was unfortunately not clearly visualized.
Video 1 Subxiphoid view from TTE

TEE was subsequently performed to rule out aortic dissection.
Video 2 TEE with a clear visualization of intimal flap in ascending aorta

Video 3 TEE with an intimal flap protruding to aortic valve cusps

Figure 2 CT examination confirmed type A aortic dissection

Patient was immediately transferred to the operating theatre, where he successfully underwent resection of ascending aorta dissection with substitution of Vascutek No. 28 vascular prosthesis.
Clinical context
Aortic dissection is a life-threatening condition originating from an intimal tear of aorta resulting in separation of the aortic wall layers and subsequent formation of a true and false lumen. This can lead to an aortic rupture (if there is an adventitial disruption) or to a second intimal tear and re-entrance into the aortic lumen. The dissection can be either antegrade or retrograde.
The classification of AD is based on anatomical location by Stanford and the older De-Bakey system.

https://www-uptodate-com.ezproxy.is.cuni.cz/contents/clinical-features-and-diagnosis-of-acute-aortic-dissection?search=aortic%20dissection&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1#H9438630
Clinical presentation and complications
- Chest pain that is abrupt and usually sharp, ripping, tearing or knife-like. Type A mostly presents with anterior chest pain, while type B frequently presents with pain in the back or abdomen. The pain may also migrate following the dissection path.
- Pulse deficit – up to 30% of patients, limb ischaemia is rare.
- Aortic regurgitation – from murmur to acute heart failure and cardiogenic shock.
- Myocardial ischaemia - as a result of false lumen expansion and coronary ostia obliteration or propagation of the dissection into the coronary tree. Typical MI ECG features.
- Pericardial effusion/tamponade (as also seen in our patient) - most commonly, the transudation of fluid across the thin wall of an adjacent false lumen into the pericardial space leads to a haemodynamically insignificant pericardial effusion, which is present in one third of patients. In some cases, rupture of the ascending aortic dissection into the pericardium can cause a rapid pericardial effusion resulting in cardiac tamponade which is frequently fatal.
- Syncope – an important symptom that is often related to life-threatening complications, such as cardiac tamponade or supra-aortic vessel dissection.
- Hypotension - may result from aortic rupture, acute severe aortic regurgitation, extensive myocardial ischaemia, cardiac tamponade, preexisting left ventricular dysfunction, or major blood loss.
- Neurological symptoms – often dramatic and masking the underlying cause – coma/stroke
- Renal failure – in up to 20% of patients, it results from renal hypoperfusion or infarction.
Learning points
- What are the risk factors for aortic dissection?
- Systemic hypertension
- Connective tissue diseases – Marfan syndrome
- Family history of aortic disease
- Known aortic valve disease
- Known thoracic aortic aneurysm
- Previous cardiac surgery with aortic manipulation
- High-speed traumas of the chest
- D-dimers level may help with diagnosis as they are typically immediately high and have high diagnostic value in the first hour. If the clinical probability is low, negative D-dimers level should rule out the diagnosis.
- Confirmation of the diagnosis requires cardiovascular imaging.
- The entire aorta must be assessed – its diameter, shape and extent of a dissection membrane, the involvement in a dissection process of the aortic valve, aortic branches, the relationship with adjacent structures, and the presence of mural thrombus.
Variety of imaging methods may be used:
- TTE – initial imaging investigation, useful for Type A AD diagnosis and as a gateway to other methods.
- TOE – is preferred in unstable patients, and can be used to monitor changes in-theatre and in post-operative intensive care
- CT - provides specific, precise measurements of the extent of dissection. The most commonly used method for AD evaluation.
- In stable patients – MRI may be performed for the most detailed image (rarely used).
- CT, MRI and TOE are equally reliable methods. If imaging is initially negative, but the suspicion for AD prevails, repetitive imaging (CT or MRI) is recommended.
- How is aortic dissection treated?
- Any patient with AD should have medical therapy for pain relief and blood pressure control. Treatment of the dissected aorta itself depends on the type of AD, severity of complications and patient’s stability.
- Type A is always indicated for urgent surgical treatment (reduced 1 month mortality from 90% to 30%), but perioperative mortality remains high (25%). This usually includes ascending aorta and/or arch replacement with possible aortic valve replacement, if the dissection involves at least one of sinus Valsalva. If organ malperfusion is present, hybrid approach with percutaneous aortic or branch artery procedure should be considered.
- Type B AD is often uncomplicated and the patient can be safely stabilized under medical therapy alone, to control pain, heart rate and blood pressure.
- If the patient with type B AD has persistent or recurrent pain, uncontrolled hypertension despite full medication, early aortic expansion, malperfusion or signs of rupture (haemothorax) the AD is termed complicated and the treatment of choice is thoracic endovascular aortic repair (TEVAR). In this procedure the proximal intimal tear is closed and blood flow is redirected back to the true lumen, leading to decompression and better distal perfusion. Thrombosis of the false lumen induces aortic remodelling and prevents aneurysmal degeneration.
- Surgery of complicated type B AD has been largely replaced by endovascular therapy, but may still be considered in patients with severe lower limb artery disease.
- If you have a patient with suspected aortic dissection
- Admit the patient to an intensive care unit as soon as possible
- Confirm the diagnosis with vascular imaging
- Control of pain
- Reduce systolic blood pressure to 100-120 mmHg
- Type A – ascending aorta is involved > usually urgent surgery
- Type B – descending or distal aorta is involved > medical treatment (nitrates and β-blockers), if complicated consider TEVAR or surgery
References
- Erbel R, Aboyans V, Boileau C, Bossone E, Bartolomeo RD, Eggebrecht H, Evangelista A, Falk V, Frank H, Gaemperli O, Grabenwöger M, Haverich A, Iung B, Manolis AJ, Meijboom F, Nienaber CA, Roffi M, Rousseau H, Sechtem U, Sirnes PA, Allmen RS, Vrints CJ; ESC Committee for Practice Guidelines. 2014 ESC Guidelines on the diagnosis and treatment of aortic diseases: Document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. The Task Force for the Diagnosis and Treatment of Aortic Diseases of the European Society of Cardiology (ESC). Eur Heart J. 2014 Nov 1;35(41):2873-926.
- Dr. Manish Barman, FESC. “Acute Aortic Dissection.” European Society of Cardiology, 2014, www.escardio.org/Journals/E-Journal-of-Cardiology-Practice/Volume-12/Acute-aortic-dissection.
- Gawinecka J, Schönrath F, von Eckardstein A. Acute aortic dissection: pathogenesis, risk factors and diagnosis. Swiss Med Wkly. 2017 Aug 25;147:w14489. doi: 10.4414/smw.2017.14489. PMID: 28871571.
- ., James H Black, III, MDWarren J Manning, MD. “Clinical Features and Diagnosis of Acute Aortic Dissection.” UpToDate, 2020, www-uptodate-com/contents/clinical-features-and-diagnosis-of-acute-aortic-dissection?search=aortic%20dissection&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1#H9438630
Authors: Michal Pazderník, Lucie Mayerová
You Might Also Like
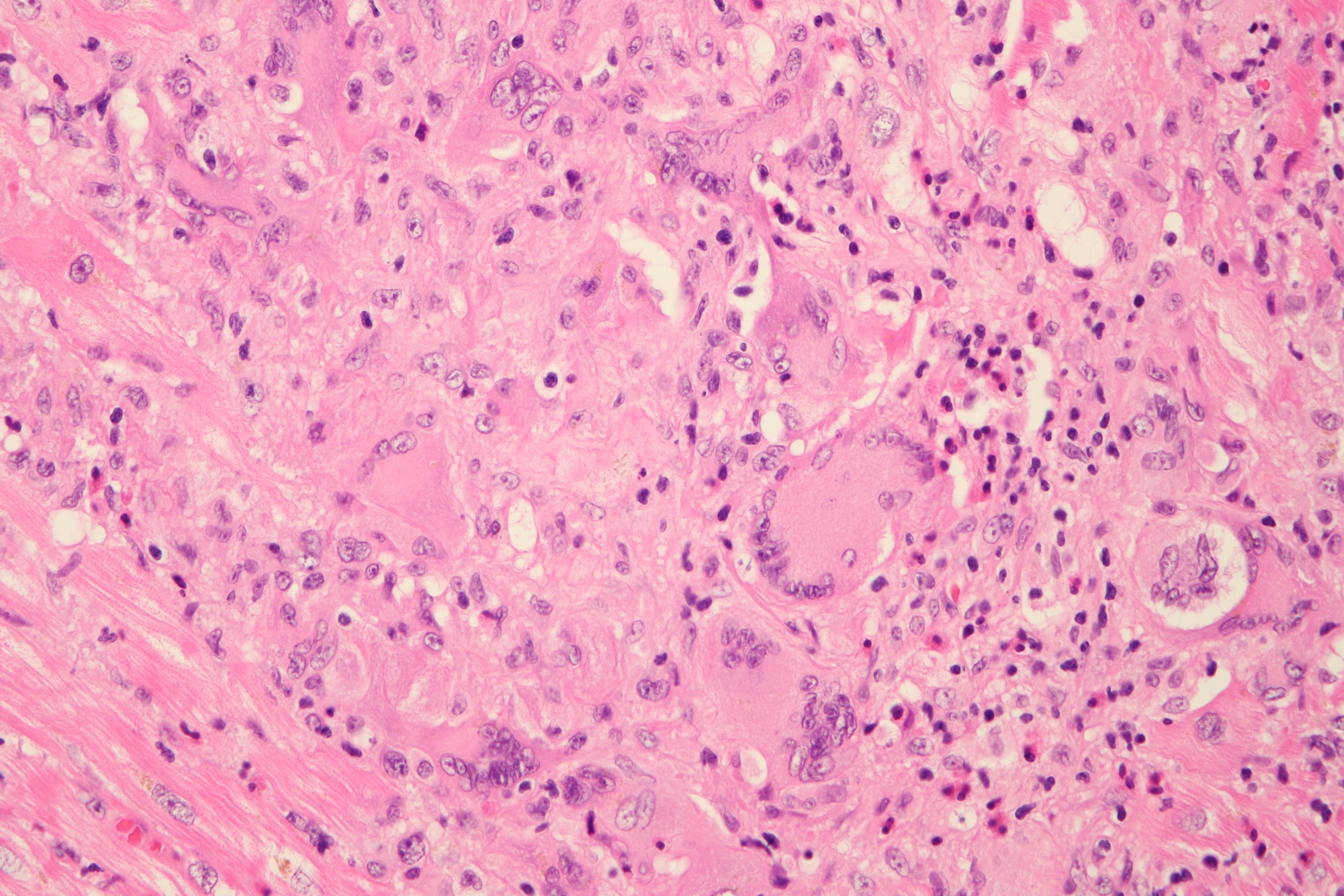
Cardiogenic shock due to Giant Cell Myocarditis
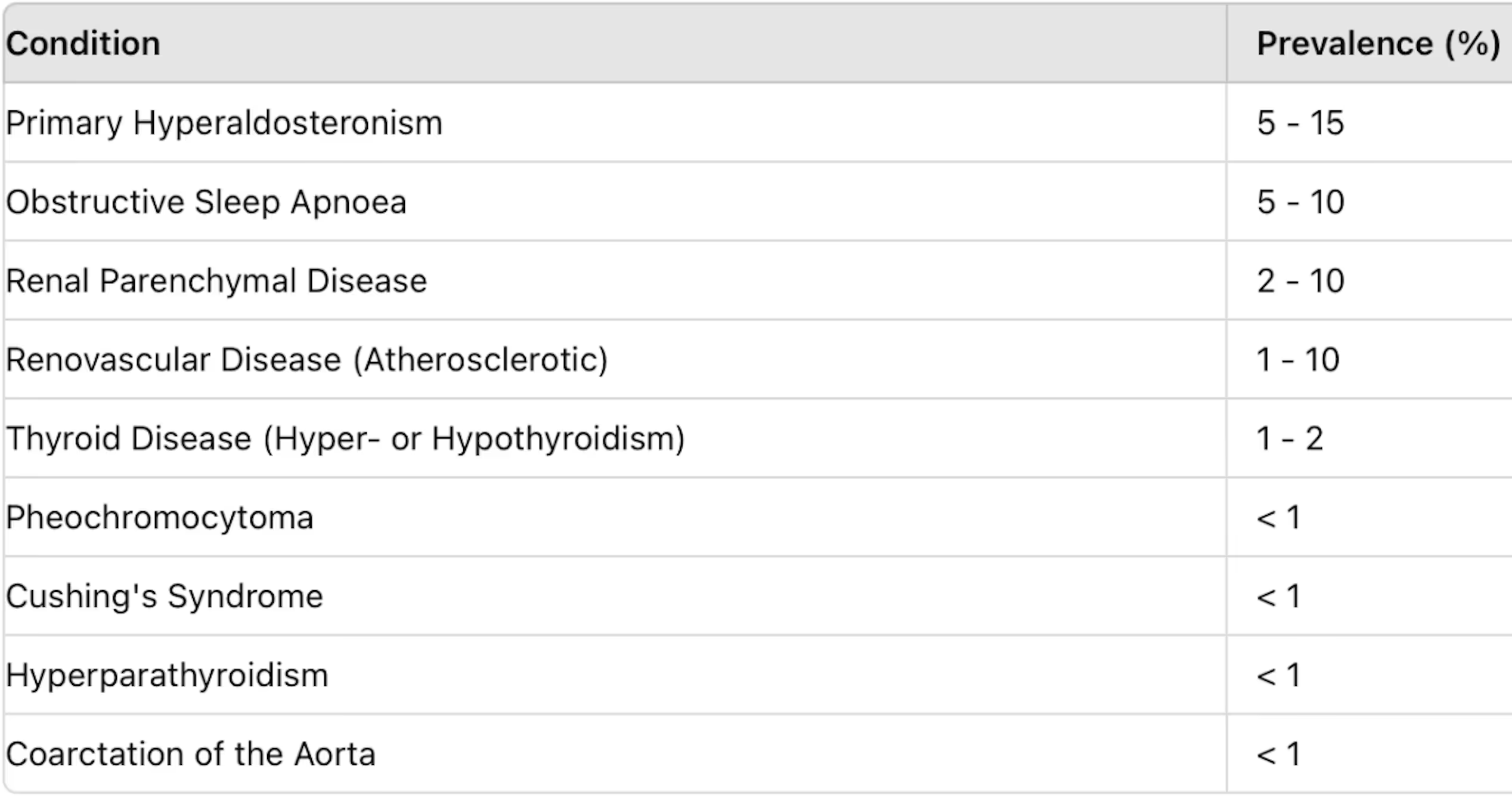
Secondary hypertension


.jpg)


