A 49-year-old patient underwent a mitral valve repair with triangular resection of the posterior leaflet and mitral valve annuloplasty with artificial ring.
2 weeks later, he presented with exertional dyspnea, fatique, cough and palpitations.
Upon admission he had a pale skin and prominent systolic murmur with it’s maximum above the apex was documented. Per rectum examination didn‘t reveal any gastrointestinal bleeding.
Laboratory tests revealed:
- hemoglobin level of 65 g/l
- MCV 84.3 fl
- schistocytes 30 (normal range 0-10/1000 RBC)
- total billirubin level = 24.8 µmol/l (normal range 3,4-20)
- free Hb = 493 mg/L (normal range <6)
- LDH = 46.4 µkat/l (normal range = 2.1-3.7)
- haptoglobin = <0,1 g/L (normal range = 0,3-2)
Direct and indirect Coombs testing was negative.
Other important laboratory tests revealed elevated urea 14.6 mmol/l and kreatinin 413.3 µmol/l suggestive of renal insufficiency.
The chest X-ray didn’t show any major pathology.
TTE showed normal ejection fraction of the left ventricle with with no regional kinetics abnormalities. The mitral valve examination showed mitral valve annuloplasty with 3-4/4 regurgitation and it also showed prolapse of the posterior leaflet with excentric jet towards interatrial septum.
The TEE examination confirmed severe 4/4 mitral regurgitation (grade 4).
Video 1 Relative prolapse of mitral valve posterior leaflet - TEE
.gif)
Video 2 Eccentric mitral regurgitation - TEE
.gif)
Video 3 3D TEE of repaired mitral valve with marked non-coaptation
.gif)
Image 1 Non-coaptation gap - 5 mm

The patient underwent gastroscopy and abdominal sonography neither of which proved a source of bleeding that could explain patient’s anemia.
The findings were suggestive of hemolytic anemia. In the presence of a heart murmur a diagnosis of intravascular hemolysis secondary to mitral valve repair failure was made.
The patient recieved red blood cell transfusions, the hemoglobin levels reached 98 g/l after which patient underwent a mitral vave replacement.
One month after cardiac surgery, no signs of hemolysis was present - normal free Hb, normal LDH, normal level of schistocytes = 8, Hb=125 g/L, kreatinin = 84 µmol/l.
Clinical context
Patophysiology of hemolysis in mitral valve surgery
Hemolysis after valve repair is either due to the failure of the repair resulting in postoperative valvular leakage or to collision of the regurgitant blood with one of the structures of the valvular apparatus
The hemolytic process has been ascribed to traumatic fragmentation of erythrocytes against prosthetic material or disruption from paravalvular jet shear forces.
Various mechanisms have been proposed as the cause for hemolysis in these patients:
1) “whiplash motion” of disrupted sutures
2) dehisced annuloplasty ring producing para-ring regurgitant jets
3) protruding paravalvular suture material or pledgets that provide an impact site against circulating red blood cells
4) delayed or insufficient endothelialization of foreign materials such as sutures or rings
5) turbulent regurgitant jet with direction against the left atrial wall.
All of these mechanisms cause traumatic fragmentation of erythrocytes and they also delay endothelialization, maintaining hemolysis.
Although shear stress contributes significantly to the pathophysiology of hemolysis, other variables may also have an effect, in particular, the type of prosthetic materials and the surfacearea of exposure.
Also some authors believe that interindividual red blood cell fragility might play a part in hemolysis, as well as an auto-immune phenomenon, as indicated by some reports of successful treatment with corticosteroids.
How to recognise hemolytic anemia after mitral valve repair?
The diagnosis of hemolytic anemia caused by mitralvalve repair is made based on clinical manifestation, laboratory findings, and other examinations to rule out or to confirm other possible reasons of anemia.
Clinical manifestation
Hemolytic anemia can preset with shortness of breath, fatigue, dizziness, pale skin, headache, confusion and jaundice.
In physical examination the finding of systolic murmur above the apex propagating towards axilla is a sign of remanifestating mitral regurgitation.
Haemolysis and renal insufficiency
Intravascular haemolysis of any cause can produce acute tubular necrosis, due to haemoglobinuria.
It presents with red/brown urine and plasma, low haptoglobin, elevated LDH, deteriorated renal function and fractional excretion of sodium less than 1%. The incidence is unknown, reaching 50% in massive haemolysis.1,2
Haemoglobin is released to the plasma, binds to haptoglobin and is degraded by the reticuloendothelial system.
When the haptoglobin is saturated, the free haemoglobin goes from its usual tetrameric form to a dimeric form. It filters through the glomerulus and goes inside the proximal tubule when it binds to the apical surface of megalin-cubulin receptor. It is there that globin and haem group are dissociated. The intracellular increase of proteins in the haem group produces nephrotoxicity caused by renal hypoperfusion, direct cytotoxicity and formation of intratubular casts interacting with Tamm-horsfall protein, which obstruct the tubules.
During massive haemolysis, deleterious effects of the nitric oxide depletion are observed: smooth muscle tone imbalance, vascular constriction, thrombosis and intrarenal vasoconstriction.
Laboratory findings
Laboratory findings of hemolytic anemia are:
1) Low levels of hemoglobin, hematocrit and red blood cells count. The mean corpuscular volume (MCV) can be normal, lower or even higher.
2) The reticulocyte index ussually exceeds 3.
3) Increased markers of red blood cell breakdown - unconjugated billirubin, free Hb and LDH
4) Decreased levels of hemopexin and haptoglobin.
5) To distinguish autoimmune process responsible for the hemolysis the direct and indirect Coombs test should be performed - in haemolytic anemia Coombs tests are negative, in autoimmune conditions are positive.
6) The peripheral blood smear should be evaluated to identify abnormal red blood cell morphologies as schistocytes, fragmented cells and polychromasia.
7) In urine analysis the markers of hemolysis are hemosiduria, hemoglobinuria and urobilinogen.
Other investigation: If indicated other examination as gastroscopy, stool testing for occult bleeding can be executed to rool out other causes of anemia.
Echocardiography evaluation
Echocardiographic study is an important tool because it allows investigation of the regurgitant jet, as well as its origin and it offers evaluation of possible mechanism of hemolysis.
The speed, direction, and impact location should be considered inthe regurgitant jet. Distinct patterns of mitral regurgitant flow disturbances have been identified in patients with mitral valve repair hemolysis. These include fragmentation, collision, acceleration, deceleration, and a free jet.
The severity of hemolysis doesn‘t appear to correlate well with the severity of regurgitation, but correlates better with the degree of shear stress created during regurgitation of the jet.
Treatment
Final treatment usually involves re-repair or MV replacement.
Conclusion
Clinically significant hemolysis is a very rare complication after mitral valve repair.
When it occurs, the best diagnostic approach is establishing the relationship between the mitral valve surgery and hemolysis using clinical examination, laboratory findings and echocardiography.
The first choice treatment remains surgical repair or replacement after the patient’s stabilization using blood transfusion or RBC masses if needed.
The surgery is needed because hemolysis rarely resolves spontaneously.
References
[1] ABOURJAILI, Georges, Estelle TORBEY a Taher ALSAGHIR. Hemolytic anemiafollowing mitral valve repair: A case presentation and literature review[online]. 2012 Winter [cit. 2021-9-9]. Dostupné z: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3627285/?fbclid=IwAR1jt6otXmS20A6AKs6vUixJ12dMvTpnTHvbuOxp3nWTjLD1OGJXAPvG1D8
[2] Cleveland Clinic. Mitral Valve Repair [online]. [cit. 2021-9-9].Dostupné z: https://my.clevelandclinic.org/health/treatments/17240-mitral-valve-repair
[3] Cardoso, C., Cachado, P. & Garcia, T.Hemolytic anemia after mitral valve repair: a case report. BMC ResNotes 6, 165 (2013). https://doi.org/10.1186/1756-0500-6-165
[4] 123sonography authors. Jet direction and mechanism of MR [online].[cit. 2021-9-9]. Dostupné z: https://123sonography.com/ebook/jet-direction-and-mechanism-mr
[5] SIGHT authors. Reticulocyte: Definition, Count, Index & Normalranges [online]. [cit. 2021-9-9]. Dostupné z: https://www.sightdx.com/knowledge-center/reticulocyte
[6] SOTANANUSAK, T, T YINGCHONCHAROEN a S CHAIYAROJ. Unlocking the mysteryof haemolytic anaemia after mitral valve repair [online]. 17 January 2020[cit. 2021-9-9]. Dostupné z: doi: https://doi.org/10.1093/ehjci/jez319.267
[7] GUNGUNES, Askin, Ibrahim AKPINAR a Mehmet DOGAN. Mechanismsof CaseReportDo All Hemolytic Anemias That Occur After Mitral Valve RepairRequire Surgical Treatment?With Mitral Prosthetic Regurgitation [online].2007. [cit. 2021-9-9]. Dostupné z: https://onlinelibrary.wiley.com/doi/pdf/10.1002/clc.20393
[8] Rosado Rubio C, Fraile Gómez P, García Cosmes P, Díez Bandera R, González Álvarez C. Intravascular haemolysis and renal failure. Nefrologia. 2011;31(5):621-2. English, Spanish. doi: 10.3265/Nefrologia.pre2011.Jun.10941. PMID: 21959740.
Authors: Michal Pazderník and Anna Marie Kubištová
You Might Also Like
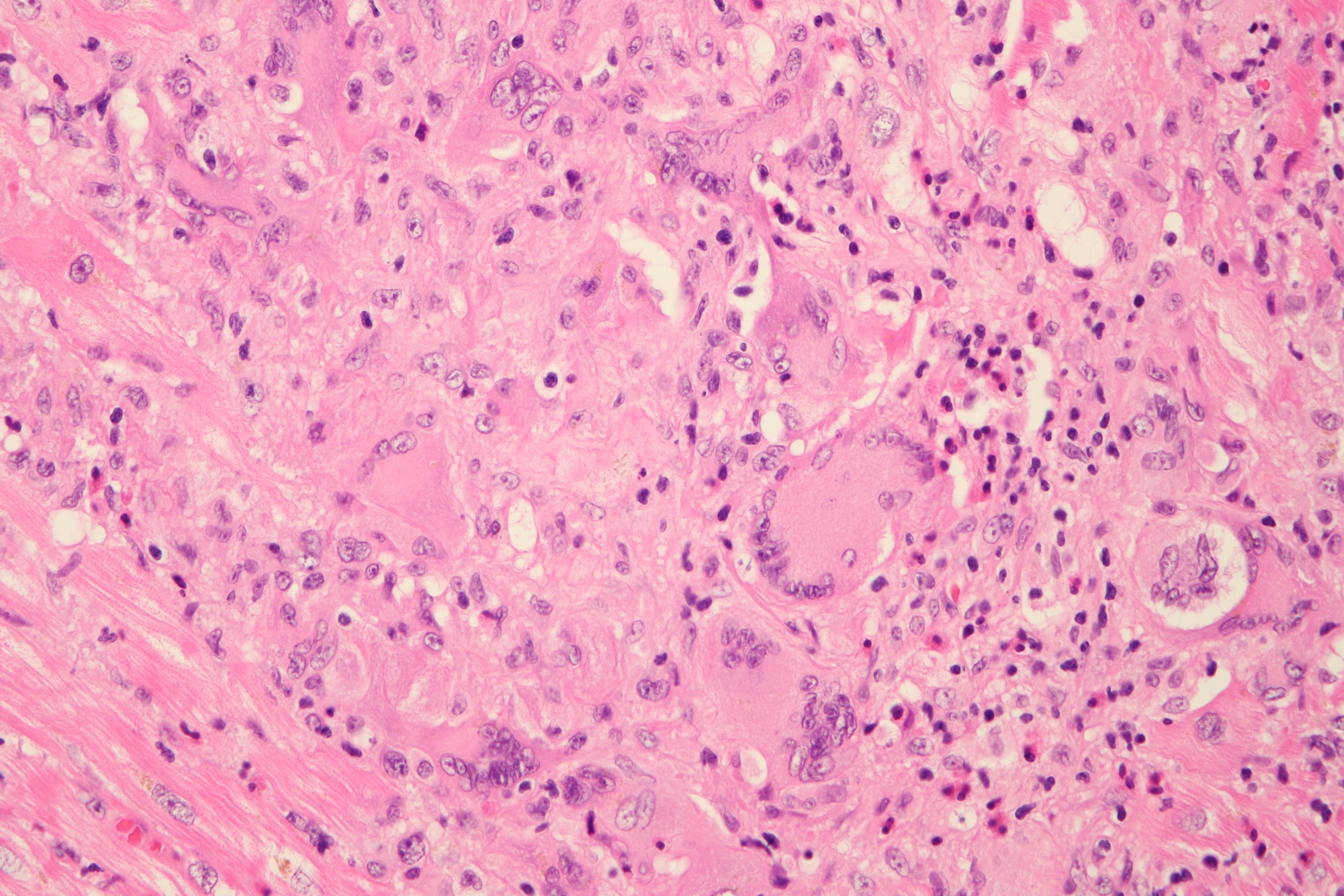
Cardiogenic shock due to Giant Cell Myocarditis
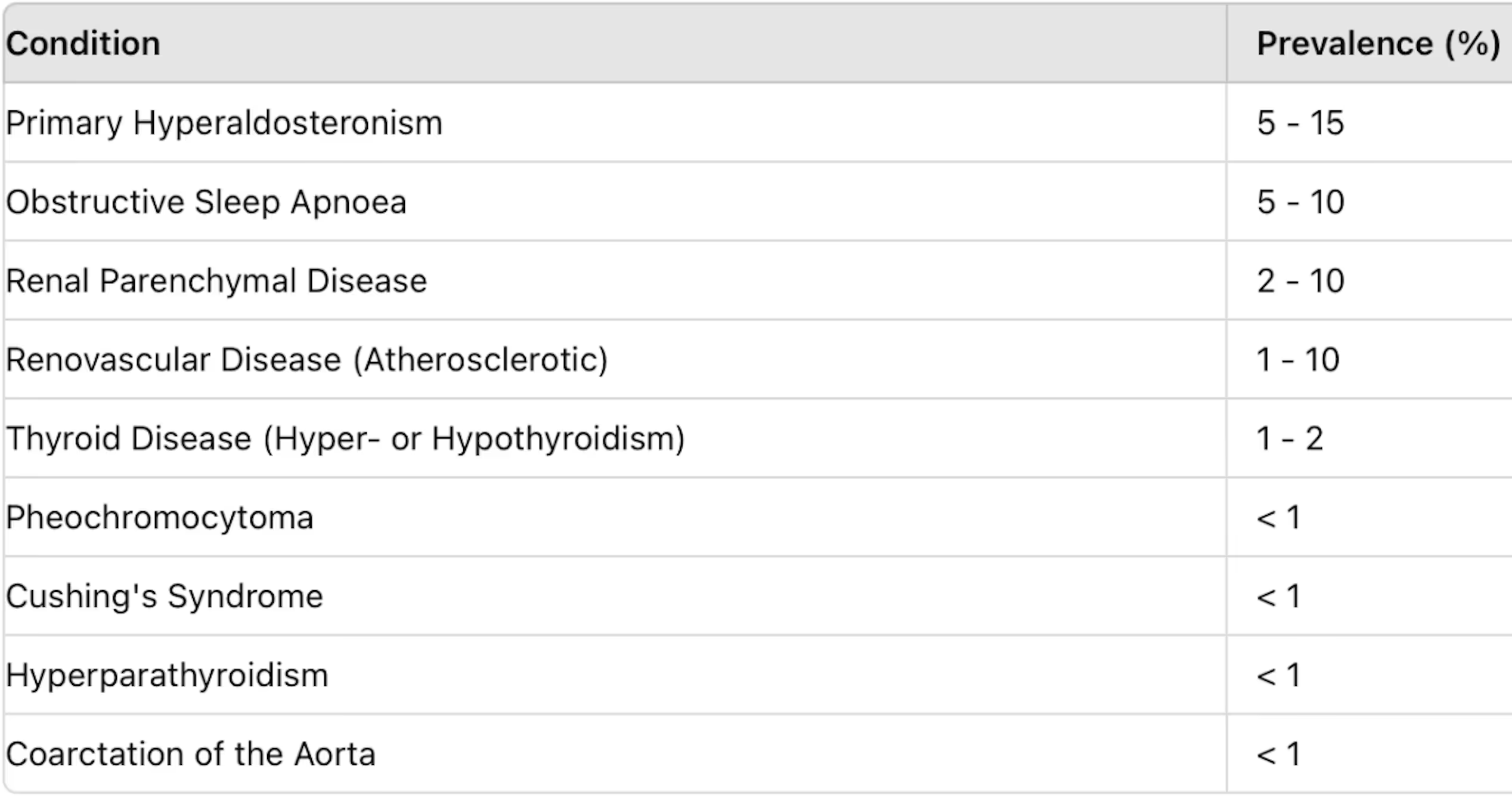
Secondary hypertension


.jpg)


