
Feochromocytoma as an unusual cause for refractory ventricular fibrillation
We present a case of 57-year-old male with a history diabetes mellitus type 2, arterial hypertension, paroxysmal atrial fibrillation and aortic valve replacement with mechanical prosthesis after an episode of infective endocarditis (25 years ago).
He was admitted to our institution after prolonged out-of-hospital cardiopulmonary resuscitation (CPR) for recurrent ventricular fibrillation (10 external DC shocks delivered in 30 minutes)
Figure 1 ECG stripes documenting 10x DC shocks delivered in the acute phase

Immediately after admission, therapeutic hypothermia was initiated and continued for 24 hours.
Bed-side transthoracic echocardiography showed moderate-to-severe left ventricular systolic dysfunction (LVEF 35%, with diffuse wall motion abnormality). Mechanical aortic prosthesis was without dysfunction and no other pathology was found.
Video 1 TTE examination on admission

Coronary angiography did not reveal any majo rpathology in coronary vessels. Brain-CT scan was normal. Toxicology laboratory results were completely negative and the most remarkable laboratory finding was severe hyperglycemia (35 mmol/l).
Patient showed signs of multiple organ dysfunction syndrome that reversed within few days at the Intensive care unit, allowing extubation on the third in-hospital day. Despite prolonged CPR (ROSC>30 minutes), patient survived without any neurological or cognitive deficit (CPC 1). For persisting signs of renal insufficiency and pancreatic irritation, abdominal ultrasonography was indicated. The dominant finding was pathological mass in the right adrenal gland. Plasma normetanephrine and chromogranin A levels and normetanephrine waste in 24-hour urinary collection were highly elevated (Figure 2).
Figure 2 Laboratory findings

Abdominal CT scan and MIBG scintigraphy confirmed hormonally active pheochromocytoma of the right adrenal gland.
Figure 3 CT image of pheochomocytoma in right adrenal gland

Figure 4 Merge of CT and MIBG scintgraphy

Figure 5 MIBG accumulation in pheochromocytoma

These findings suggested an association between recurrent ventricular fibrillation and excessive catecholamine secretion. However, there were no symptoms and signs suggestive of pheochromocytoma till resuscitation. After an exclusion of metastatic gastrointestinal tumor, implantable cardioverter-defibrilator (ICD) was implanted to provide secondary prevention of sudden cardiac death. Echocardiography performed two weeks later documented normalization of the LV systolic function (LVEF 55-60%), suggesting initial post-resuscitation systolic dysfunction. One month later, after stabilization and pharmacological preparation, patient underwent right-sided adrenalectomy (laparoscopic approach was not possible due to obesity and anatomical conditions).
Figure 6 Pheochormocytoma after resection

Levels of metanephrines in blood and urine normalized after the surgery. In addition, better compensation of diabetes mellitus and arterial hypertension was achieved.
Clinical context
1) Which laboratory markers may be used for diagnosis of pheochromocytoma?
Catecholamines produced by pheochromocytoma are metabolized within thechromaffin cells. Norepinephrine is metabolized to normetanephrine and epinephrine to metanephrine. Since this process occurs within the tumor, independently of catecholamine release, pheochromocytomas are best diagnosed by assessment of these metabolites. Plasma metanephrine testing has the highest sensitivity (96%) for detecting pheochromocytoma, while it has lower specificity (85%). In comparison, 24-hour urinary collection of catecholamines and metanephrines has a sensitivity of 87.5% and a specificity of 99.7%.
Another diagnostic possibility is detection of chromogranin A. It is an acidic monomeric protein that is stored and secreted together with catecholamines. Plasma levels of chromogranin A were reported to 83% sensitive and 96% specific for identification of pheochromocytoma [3,4] .
2) What is optimal treatment of pheochromocytoma and what should we notforget to do before operation?
The only curative method that allows complete resolution of symptoms is complete resection of the tumor. Careful preoperative management is required to control blood pressure, correct fluid volume, and prevent intraoperative hypertensive crises. Alpha blockade should be initiated 10-14 days preoperatively to allow for expansion of blood volume. The patient should undergo volume expansion with isotonic sodium chloride solution. Beta blocker therapy should be started after adequate alpha blockade (usually after 2 days). If beta blockade is started prematurely, unopposed alpha stimulation could precipitate a hypertensive crisis. Since the first laparoscopic adrenalectomy was performed in 1992, this approach has been adopted quickly as the procedure of choice. It is widely accepted that the laparoscopic approach provides better visualization of anatomically complex areas through smaller and less painful incisions. This approach is also associated with lower complication rates, less operative blood loss, less postoperative pain and earlier return to activity. The hospital stay is sporter and overall costs lower. In addition, is provides better cosmetic results. On the other hand, laparoscopic adrenalectomy should be performed only when it can achieve complete tumor resection with an intact adrenal capsule. This is very important, as complete resection constitutes the only possibility of curing patients, and it should be taken into account that even small tumors may rarely be malignant. Conversion to an open procedure should be an early decision, prior to disruption of the tumor capsule. The conversion (about 5%) to open surgery is recommended for large lesions(>8 cm) with the suspicion of malignancy [5,6,7]. Also in our patient, the initial laparoscopic procedure was converted into classical open surgery due to hepatomegaly, obesity and contactly bleeding fragile tissues.
3) What is the reason for episodes of excessive hyperglycemia or worse compensation of diabetes mellitus in pheochromocytoma patients?
There are several mechanisms for hyperglycemia in pheochromocytoma patient. Many of these patients have decreased glucose tolerance, probably due to catecholamine-induced low insulin production. Another explanation is that surges in epinephrine levels cause corresponding release of glucagon - which stimulates the production and release of glucose - while inhibiting insulin utilization. Increased release of glucagon further stimulates catecholaminesurges and may lead to ketoacidosis.
4) What are the symptoms of elevated catecholamines levels?
The reaction of the organism to excess catecholamines levels is very individual. On one side, there could be fully asymptomatic patients. Most patients have typical symptoms such as palpitations, cefalea, sweating or nervousness, associated with episodes of hypertension. However, totally atypical presentations can occur-such as intestinal pseudo-obstruction syndrome or epilepsy (described more than eighty different types of expressions). Occasionally, patients with predominantly epinephrine-secreting tumours present with hypotension or even shock. Other cardiovascular complications of pheochromocytoma include ischaemic heart disease, acute myocardial infarction (with normal coronarogram), cardiac arrhythmias, heart failure due to toxic cardiomyopathy, or pulmonary edema. Catecholamines have been shown to influence the extracellular matrix with collagen deposition and subsequent fibrosis in the arterial wall and in the myocardium. Incidence of cardiovascular complications as an initial presentation of pheochromocytoma is relatively high (12-19%), especially in patients with large tumors and high levels of biochemical markers. Early diagnosis is mandatory to prevent severecomplications [8,9,10].
5) Is there any familiar occurrence of pheochromocytoma?
In approximately 20% of individuals, pheochromocytoma is a part of multiple endocrine neoplasia with genetic background. Family presentation in younger patients and multifocal occurrence of pheochromocytoma are typical in such cases.
6) Was an indication to implant an ICD appropriate?
On one side, electrical storm in our patient could be regarded as truly reversible. In such cases, a wearable defibrillator could be considered. On the other hand, in the setting of artificial valve and moderate-to-severe left ventricular dysfunction, the risk of recurrence of malignant arrhythmia was believed to be unpredictable. Also latent dissemination of tumor could not be excluded at thetime of manifestation. Another argument was possible recurrence of pheochromocytoma. However, the recurrence of resected malignant pheochromocytoma has been reported more often 6 and more years following curative surgery. Therefore, careful and sustained clinical and biochemical follow-up are needed.
For all the above reasons and after a thorough discussion, the patient favored implantation of an ICD. Although the indication for ICD implantation did not meet fully the existing guidelines [12], it proved to be correct. Six months after implantation, the patient presented with an episode of polymorphicventricular tachycardia (heart rate 315/min), terminated with the first shock (patient reported transient weakness without syncope).
Conclusions and implications for clinical practice
The key take-home message of this case report is that all survivors of cardiac arrest should be thoroughly evaluated in a specialized cardiovascular center. In this case, pheochromocytoma was diagnosed as the most probable explanation for intractable ventricular fibrillation. Rapid diagnosis of pheochromocytoma is based on a combination of typical clinical features, assessment of elevated level of catecholamines and their metabolites in blood and urine, followed by imaging techniques. Life prognosis depends on localization and the extent of pheochromocytoma.It varies from complete resolution of symptoms after surgery with a goodprognosis to short life expectancy in patients with malignant pheochromocytoma and metastatic dissemination. After surgery, also better compensation of diabetes mellitus and arterial hypertension could be expected.
References
1. Zelinka T. Pheochromocytoma, Internal Med. 2007; 1: 30–38
2. Alessadro Maloberti et al.,Acute coronary syndrome: A rare case of multiple endocrine neoplasia syndromeswith pheochromocytoma and medullary thyroid carcinoma, CancerBiol Med. 2015 Sep; 12(3): 255–258.
3. Pappachan JM, Diagnosis and management of pheochromocytoma: a practical guide to clinicians, CurrHypertens Rep. 2014 Jul;16(7):442
4. Grossrubatscher E et al., The role of chromogranin A in the managementof patients with phaeochromocytoma. Clin Endocrinol (Oxf) 2006. Sep;65(3):287-293
5. Conzo G. et al., Laparoscopic adrenalectomy, a safe procedure forpheochromocytoma. A retrospective reviewof clinical series. Int J Surg 2013;11(2):152-156
6. Zografos G.N. et al., Laparoscopic Surgery for MalignantAdrenal Tumors, Journal of the Society of Laparoendoscopic Surgeons 2009Apr-Jun; 13(2): 196–202
7. Barski D. Managementand follow up of extra-adrenal phaeochromocytoma. Cent European J Urol 2014;67:156-161
8. Galetta F, FranzoniF, Bernini G, Poupak F, Carpi A, Cini G, et al. Cardiovascularcomplications in patients with pheochromocytoma: a mini-review. Biomed Pharmacother 2010;64:505-509
9. Zelinka T, Petrák O,Turková H, Holaj R, Strauch B, Kršek M, et al. Highincidence of cardiovascular complications in pheochromocytoma. Horm Metab Res 2012;44:379-384
10. Yu R, Nissen NN,Bannykh SI.. Cardiac complications as initialmanifestation of pheochromocytoma: frequency, outcome, and predictors. Endocr Pract 2012;18:483-492
11. Philip C. Johnston, Ulster Med J. 2015 May;84(2): 102–106, Recurrence ofPhaeochromocytoma and Abdominal Paraganglioma After Initial SurgicalIntervention
12. Silvia Prioni et al., ESC Guidelines for the managementof patients with ventricular arrhythmias and the prevention of sudden cardiacdeath, European Heart Journal (2015) 36, 2793–2867
Authors: Pavel Hajdusek, Michal Pazdernik, Jiri Kettner, Mariya Kalantay, Josef Kautzner
You Might Also Like
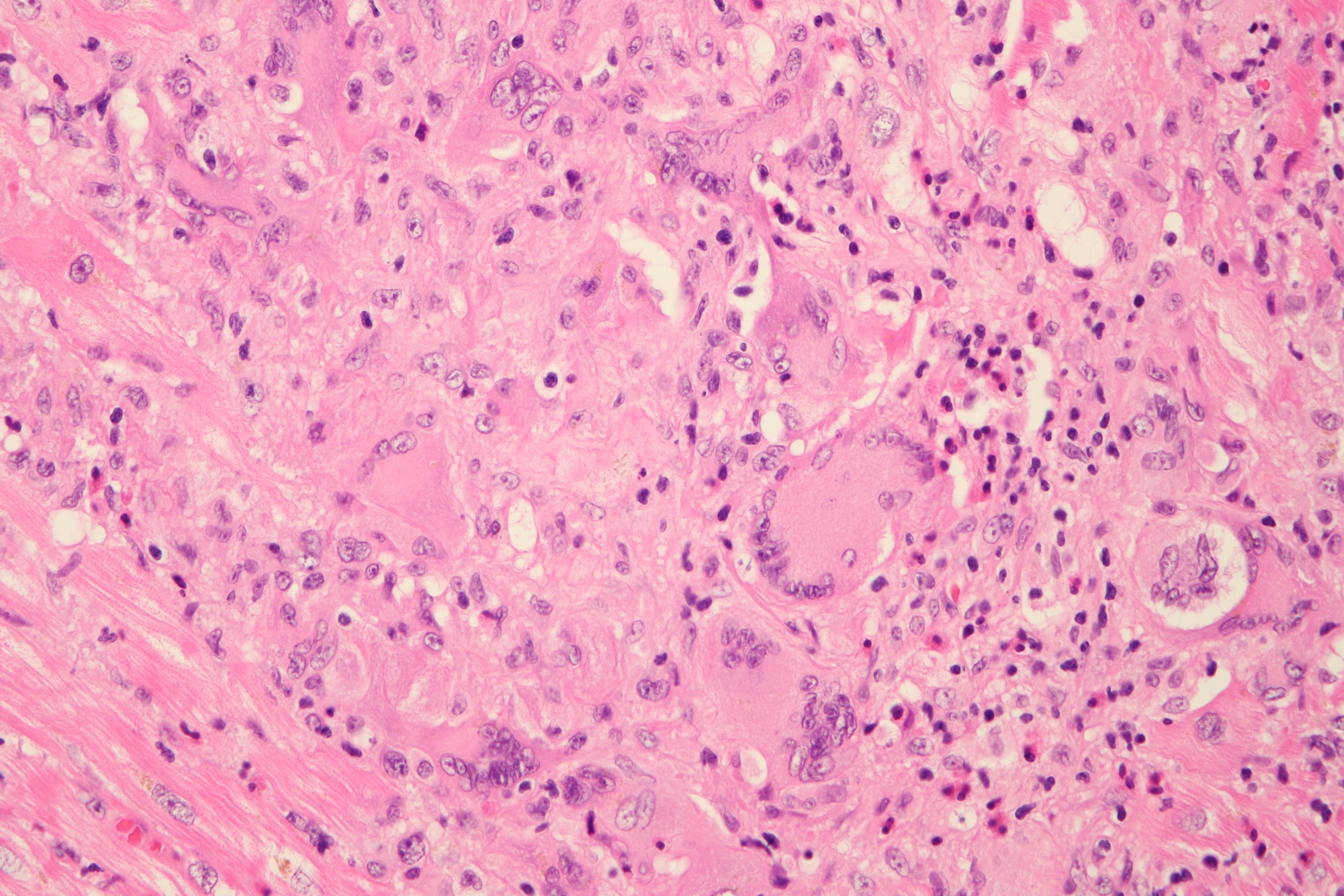
Cardiogenic shock due to Giant Cell Myocarditis
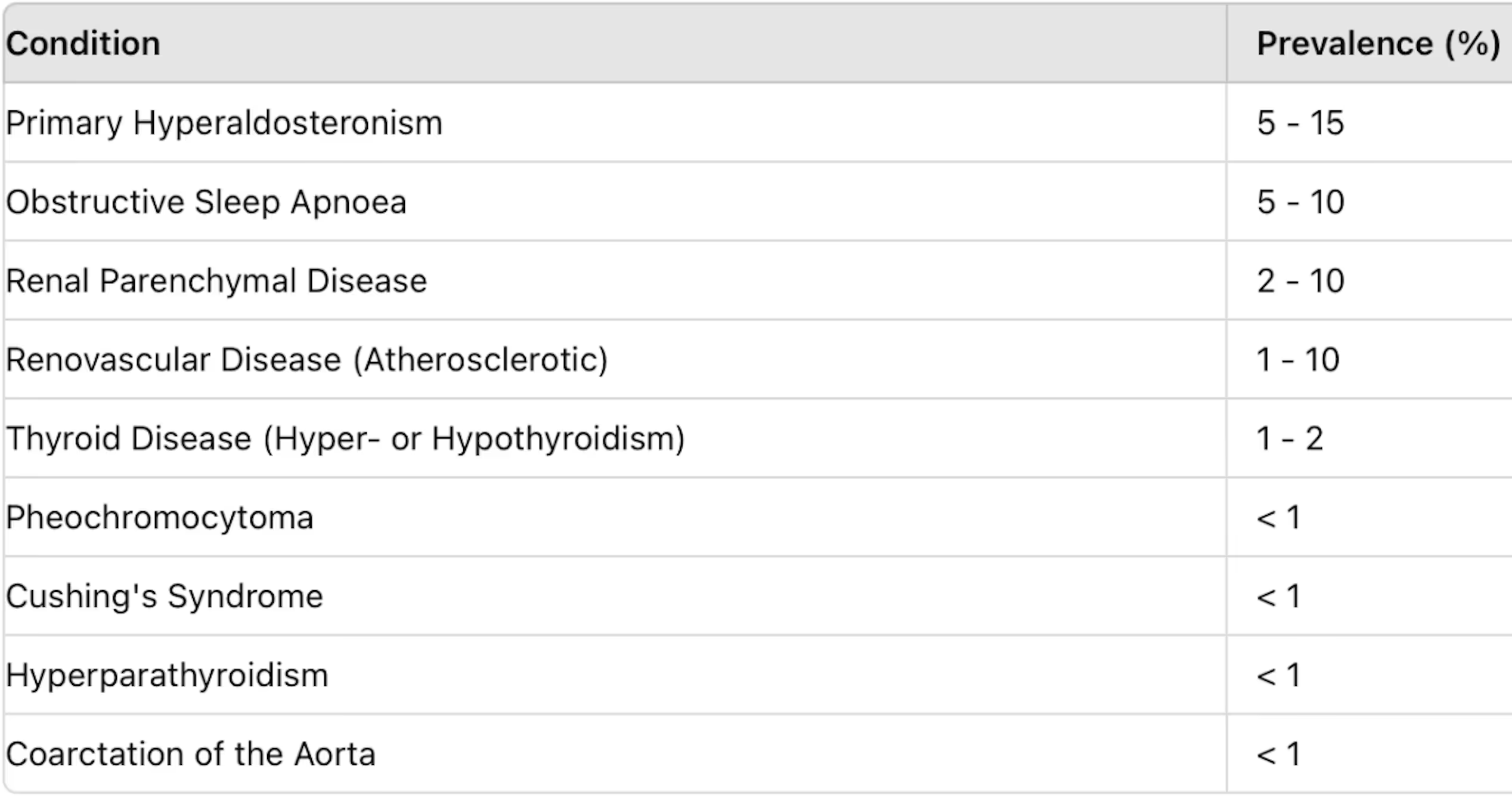
Secondary hypertension

.jpg)


